Biological causes of osteoporosis
Bones are living tissue which have their own blood vessels and are made of various cells, proteins, minerals and vitamins. This structure enables them to grow, transform and repair themselves throughout life.
We are born with about 300 soft bones. During childhood and adolescence, cartilage grows and is slowly replaced by hard bone. Some of these bones then later fuse together, resulting in an adult skeleton with 206 bones.
The major functions of bones are to:
- Provide structural support for the body
- Provide protection of vital organs
- Provide an environment for marrow (where blood cells are produced)
- Act as a storage area for minerals (such as calcium)
Bones are made of two tissue types:
- Cortical (compact) bone is the dense, hard outer layer of bones which comprises approximately 80% of total bone mass. It is organised into osteons (Haversian system) at the microscopic level. Each osteon consists of a central canal (Haversian canal) containing blood vessels, nerves, and lymphatic vessels, surrounded by concentric layers of lamellae. This structure gives the bone its stiffness and strength.
- Trabecular (cancellous, spongy) bone: Trabeculae are composed of lamellae that run largely parallel to the trabecular surface and the remnants of older, remodelled bone that may appear osteon-like in some areas. This structure provides a high surface area and is metabolically active, making it particularly sensitive to hormonal and mechanical influences.
Bones are composed of:
- Osteoblasts and Osteocytes: these are bone forming cells
- Osteoclasts: these are bone resorbing cells
- Osteoid: this is the non-mineral, organic part of the bone matrix made of collagen and non-collagenous proteins
- Inorganic mineral salts deposited within the matrix
BONE MICROARCHITECTURE
Bone microarchitecture refers to the microscopic and sub-microscopic structural organisation of bone tissue. It reflects how cortical and trabecular components are arranged, directly contributing to bone strength, quality and mechanical resistance. Microarchitecture is a major determinant of bone fragility, independent of bone mineral density (BMD). Deterioration of the microarchitecture is key in osteoporosis.
KEY MICROARCHITECTURAL PARAMETERS
The following parameters are widely used to quantify bone microarchitecture. Their deterioration is strongly associated with osteoporotic fracture risk.
Trabecular Thickness (Tb.Th)
- In healthy adult humans, trabecular bone is typically composed of plate- and rod-like structures of around 200 µm thickness.
- Osteoporosis leads to thinning of the trabeculae, reducing the width of trabecular plates and rods.
- Thinner trabeculae are more prone to microdamage and contribute less to load distribution.
Trabecular Number (Tb.N)
- Represents the number of trabeculae per unit length.
- Healthy bone has a dense, interconnected network where horizontal trabeculae brace vertical trabeculae.
- Osteoporosis can cause the complete loss of some trabeculae, not merely their thinning, resulting in:
- A reduction in the number of trabeculae
- Structural discontinuities
- Loss of horizontal trabeculae that stabilise vertical load-bearing elements
Trabecular Separation (Tb. Sp)
- Measures the average distance between the trabeculae.
- As the trabeculae thin and disappear, the separation increases.
- Increased Tb. Sp is strongly associated with vertebral fragility and decreased mechanical strength.
Trabecular Bone Volume Fraction (BV/TV)
- Ratio of trabecular bone volume to total volume
- A key determinant of overall trabecular strength
- Significantly reduced in osteoporosis, reflecting both thinning and loss of trabeculae.
Cortical Thickness (Ct. Th)
- Reflects the width of the cortical shell.
- Thinning occurs with age and accelerates in cases of osteoporosis.
- The reduction of cortical thickness is a key indicator of bone loss and high risk of fracture.
- A marker of bone quality.
Cortical Porosity (Ct. Po)
- Measures the proportion of cortical bone occupied by pores (void spaces).
- Increases with age, menopause, and osteoporosis.
- Elevated cortical porosity is a key indicator of weaker, more fragile bone.
MICROARCHITECTURAL DETERIORATION IN OSTEOPOROSIS
Osteoporosis affects bone not only by reducing mineral density but also by degrading its internal structure. Key changes include:
- Trabecular thinning
Reduces load-bearing capacity and increases susceptibility to microdamage.
- Loss of connectivity
- Horizontal trabeculae are preferentially lost.
- The remaining structure becomes discontinuous.
- Vertical trabeculae are more prone to buckling under load.
- Plate-to-rod conversion
Healthy trabecular bone is dominated by plate-like structures, which provide superior mechanical strength. Osteoporosis leads to:
- Loss of plates
- Conversion into rod-like trabeculae
- Reduced resistance to bending and compression`
- Cortical porosity
Although primarily a trabecular disease, osteoporosis also increases cortical porosity and reduces cortical thickness, contributing significantly to hip and wrist fracture risk.
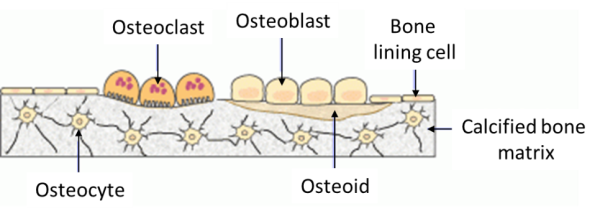
Bone Cells
Cells in our bones are responsible for bone formation, resorption, maintenance and (re-)modelling:
Osteoblasts: These cells are derived from mesenchymal stem cells and are responsible for bone matrix synthesis and its subsequent mineralization. In the adult skeleton, the majority of bone surfaces that are not undergoing formation or resorption (i.e. not being remodelled) are lined by bone lining cells.
Osteocytes: These cells are osteoblasts that become incorporated within the newly formed osteoid, which eventually becomes calcified bone. Osteocytes situated deep in bone matrix maintain contact with newly incorporated osteocytes in osteoid, and with osteoblasts and bone lining cells on the bone surfaces, through an extensive network of cell processes (canaliculi). They are thought to be ideally situated to respond to changes in physical forces upon bone and to transduce messages to cells on the bone surface, directing them to initiate formation or resorption responses.
- Osteoclasts: These cells are large multinucleated cells, like macrophages, derived from the hematopoietic lineage. Osteoclasts function in the resorption of mineralized tissue and are found attached to the bone surface at sites of active bone resorption. Their characteristic feature is a ruffled edge where active resorption takes place with the secretion of acid and bone-resorbing enzymes, which digest bone mineral and bone matrix.
Bone matrix
Osteoid is comprised of type I collagen (~94%) and non-collagenous proteins. The hardness and rigidity of bone is due to the presence of mineral salt in the osteoid matrix, which is a crystalline complex of calcium and phosphate (hydroxyapatite). Calcified bone contains about 25% organic matrix (2-5% of which are cells), 5% water and 70% inorganic mineral (hydroxyapatite).
Types of bone
Two types of bone can be distinguished based on the microscopic three-dimensional pattern of collagen forming the osteoid:
Woven bone: disorganised collagen architecture resulting in mechanically weak tissue
Lamellar bone: regular alignment of collagen into sheets (lamellae) producing strong and resilient tissue
Woven bone is produced when osteoblasts produce osteoid rapidly. This occurs initially in all foetal bones, but the resulting woven bone is replaced by remodelling and the deposition of more resilient lamellar bone. In adults, woven bone is formed when there is very rapid new bone formation, as occurs in the repair of a fracture. Following a fracture, woven bone is remodelled, and lamellar bone is deposited. Virtually all bone in the healthy mature adult is lamellar bone.
Bone development and growth
Bone development is a coordinated biological process during which the skeleton forms, matures, and acquires the structural and microarchitectural characteristics that determine its mechanical performance throughout life. This process comprises two major actions: bone formation and resorption. These actions allow bones to expand in diameter through periosteal apposition of cortical bone and to lengthen through endochondral ossification. This process of bone modelling begins during foetal development and continues throughout childhood and adolescence, determining the size, shape, and internal architecture of the adult skeleton. Bone modelling continues until the growth plates close, typically by the end of the second decade of life, after which the skeleton transitions to a remodelling-dominant state.
Bone modelling
Bone modelling refers to the process by which bone formation and resorption occur on separate surfaces, that is, these two activities are not coupled. This mechanism:
- Determines the shape of the skeleton
- Leads to an increase in the length and diameter of long bones
- Establishes the structural foundation for adult bone strength
As illustrated in the figure below, peak bone mass is generally achieved for both males and females by the mid-twenties. Thereafter, a gradual decline into old age occurs in men, while a plateau followed by an accelerated period of bone loss for several years after the menopause occurs in women.
With the size of our skeleton and the amount of bone contained in it changing significantly throughout life, overarching objectives to maintain good bone health at various stages of life are described in prevention throughout life.
Bone remodelling
Once peak bone mass has been achieved in adulthood (described above), the bone mass and structural integrity is maintained by a process called remodelling, as illustrated in the figure below.
More specifically, remodelling is the replacement of old bone tissue by new bone and continues throughout life, so that most of the adult skeleton is replaced about every 10 years. Unlike modelling, remodelling is a coupled process in which bone formation and resorption occur sequentially at the same site, consisting of different phases as shown below.
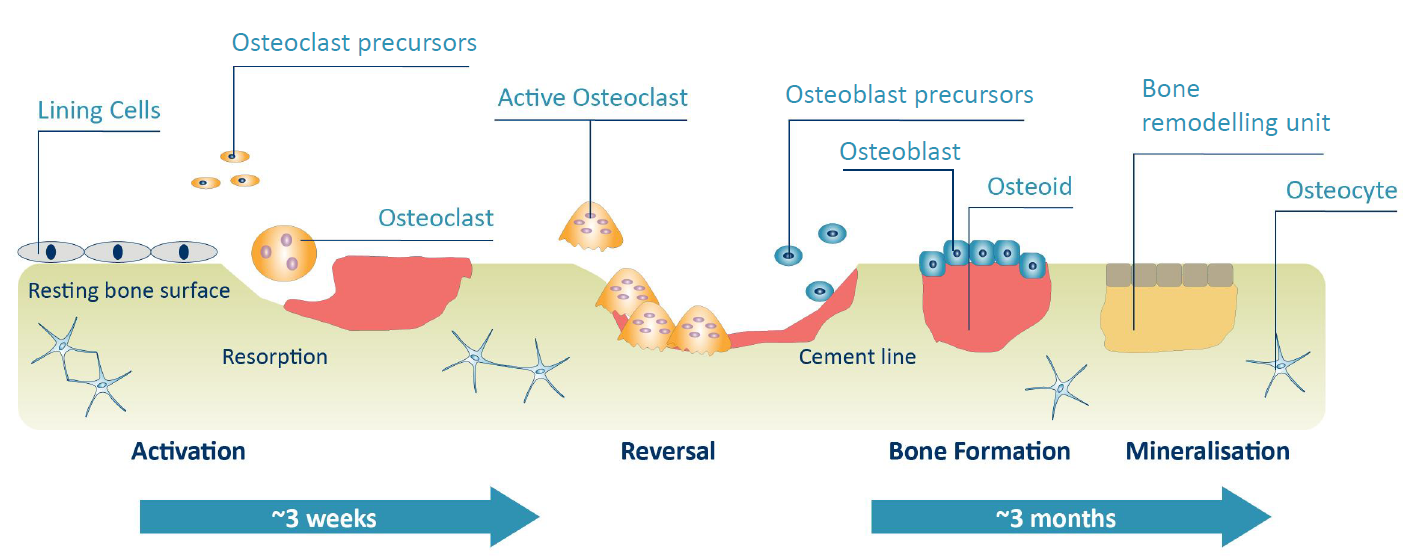
Activation: Initiated by the contraction of lining cells and the recruitment of osteoclast precursors to the remodelling site.
Resorption: Active, multinucleated osteoclasts adhere to the bone surface and remove mineralised tissue (old bone) by acidification and proteolytic digestion.
Reversal: After resorption, osteoclasts leave the site and osteoblast precursors arrive, differentiating into mature osteoblasts.
Bone formation: Mature osteoblasts cover the excavated area and begin to synthesise osteoid, thus initiating the formation of new bone.
Mineralisation: Osteoid mineralises to form new bone. Osteoblasts then flatten and transition into bone lining cells in the newly formed surface.